Editorial
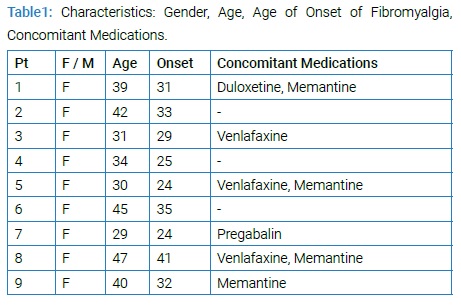
Fibromyalgia (FM) is a chronic syndrome characterized by widespread musculoskeletal pain, fatigue, stiffness, and associated features such as insomnia, mood and anxiety symptoms and cognitive dysfunction. In the general population, FM is prevalent with a rate of 2%–4% −mainly women− and causes functional impairment and disability [1]. Most patients with FM require a combination of medications and other interventions [2,3]; yet a partial response is common. FM supposedly results from the enhanced excitability of the spinal cord neurons transmitting nociceptive information to the brain. Central sensitization would explain allodynia, or pain at tender points, and hyperalgesia in FM [4]. Functional brain imaging studies have shown dysfunctional connectivity of the pain network in FM [5,6]. Neuromodulation is therefore a plausible treatment modality for FM. Transcranial Photobiomodulation (t-PBM) with near-infrared light (NIR) is an experimental, non-invasive neuromodulation technique for neuropsychiatric disorders [7]. t-PBM improves mitochondrial function, increases ATP levels, increases synaptogenesis and neurogenesis, while it reduces inflammation, edema and oxidative stress [7]. Patients with treatment-resistant FM were offered t-PBM by their physician (PM). They were informed of the possible benefits and adverse effects and agreed to off-label use. This chart-review of FM patients treated with t-PBM was approved by the Institutional Review Board of the Massachusetts General Hospital. This report describes the use of t-PBM in nine women with FM (mean age 37.4 ± 6.7; mean age of FM onset 30.4 ± 5.6), who were treated with 2 in-office sessions of t-PBM per week for 4 weeks, as an add-on to stable pharmacotherapy (Table 1).
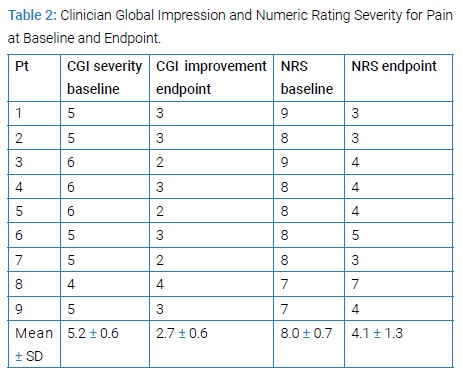
t-PBM was delivered with the Omnilux New U device (Photomedex Inc) applied to the forehead. t-PBM was delivered with the following parameters: 830 nm; 33.2 mW/cm2; 40 J/cm2; 28.7 cm2 and 20 min per site; 2 sequential sites based on electroencephalography points: F3 and F4; 2.3 kJ per session. All patients were evaluated at baseline, as part of their ongoing clinical care, and were assessed every 2 weeks by their physician for safety, tolerability, FM symptoms and pain, according to standard clinical practice. t-PBM sessions were well tolerated and no adverse experiences were reported. FM symptoms−both at baseline and after 4 weeks−were quantified by the Clinician Global Impression (CGI) severity and improvement scales, and by the Numeric Rating Scale (NRS) for pain severity, the latter rated on a scale zero “no pain” to ten “worst pain”. At baseline, the CGI-severity score was 5.2 ±0.6 (mean ± SD), reflecting at least “marked severity” of FM symptoms, despite medications. At the endpoint, the CGI-improvement score averaged 2.7 ± 0.6, showing more than “minimal improvement” of FM symptoms,and the NRS score decreased significantly from 8.0 ± 0.7 to 4.1 ± 1.3, nearly half of the original pain level (t = 6.61, df = 8, p < 0.001; (Table 2).
Clinical trials conducted in FM patients have shown the benefits of systemic PBM (s-PBM), applied at different tender points, in decreasing pain and improving quality of life [8,9]. In fact, s-PBM promotes healing in a range of musculoskeletal pathologies and induces analgesia [10]. Our report suggests that t-PBM might also have a role (alone or in augmentation) for the treatment of FM.
Acknowledgements
Dr. Cassano has received consultation fees from Janssen Research and Development and from Niraxx Light Therapeutics Inc. Dr. Cassano has received unrestricted funding from Photothera Inc. and then from Litecure Inc. to conduct studies on transcranial photobiomodulation for the treatment of major depressive disorder and a study on healthy subjects. He has also received funding from Cerebral Sciences to conduct a study on transcranial photobiomodulation for generalized anxiety disorder. Dr. Cassano cofounded a company (Niraxx Light Therapeutics Inc) focused on the development of new modalities of treatment based on near-infrared light. Dr. Cassano has filed several patents related to the use of near infrared light in psychiatry.
Conflict of interest
All other authors have no conflict of interest to declare.
Keywords
Transcranial photobiomodulation; Fibromyalgia; Neuropsychiatric disorder
Cite this article
Mannu P, Spera V, Maiello M, Cassano P. Transcranial photobiomodulation for fibromyalgia. Clin Case Rep J. 2020;1(3):1–2.
Copyright
© 2020 Paolo Cassano. This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY-4.0).


