Treatment of a Partially Necrotic Molar Tooth by Applying Apexogenesis Principles
* Mickeviciene L;
Venskutonis T;
-
* Mickeviciene L: Clinic of Dental and Oral Pathology, Faculty of Odontology, Lithuanian University of Health Sciences, Kaunas, Lithuania.
-
Venskutonis T: Clinic of Dental and Oral Pathology, Faculty of Odontology, Lithuanian University of Health Sciences, Kaunas, Lithuania.
-
Mar 17, 2023 |
-
Volume: 4 |
-
Issue: 2 |
-
Views: 6221 |
-
Downloads: 2461 |
Abstract
The most suitable endodontic treatments for immature teeth vary depending on the case selection criteria, the dental pulp’s status, the root’s maturity, and the prognosis. This report describes the case of an immature first lower molar tooth with necrotic pulp that was to be treated with the apexification method. During the endodontic procedure, the pulp sensitivity in the apical third of the canal was evaluated as positive. Therefore, the treatment was changed from apexification to modified apexogenesis. The working length was measured until the pulp sensitivity response, and the canals were chemo-mechanically prepared at that length. Then, endodontic treatment using calcium hydroxide short-term therapy and a 3-mm mineral trioxide aggregate plug in the present working length was applied. Periapical healing and apical closure were achieved during the follow-up period, and root length increased.
Introduction
Apical periodontitis only develops when necrosis and infection affect the entire pulp and reach the periodontal tissues. Several studies have revealed that the pulp can still be vital even when a periapical radiolucency is seen on the radiograph [1–4]. Partial pulp necrosis in teeth with apical periodontitis is seen clinically when the endodontic instrument provokes bleeding and pain before it reaches the apex [5,6]. This condition is more common in immature teeth. Total pulp necrosis is slowed down by the immune system’s cellular and molecular elements carried through the large foraminal opening [5,4]. Langeland et al. and Lin demonstrated that vital pulp tissue could be preserved in a tooth after periapical radiolucency has developed [7,8]. In this case, immature teeth should be treated as conservatively as manageable to allow apexogenesis to occur.
Apexogenesis maintains the vital pulp’s survival to continue root maturation and thickening, strengthening the tooth and making it more resistant to fracture [9]. Because the apical part of the pulp is still vital and able to mineralize root dentine, there is no rational justification for providing full endodontic treatment or apexification. It is important to remove the infected pulp to prevent the spread of necrosis in order to allow the remaining vital, uninfected, and uninflamed pulp to continue the root development process [10,11]. This report demonstrates an immature mandibular molar presenting with a clinical diagnosis of necrotic pulp and periapical radiolucencies. Vital tissue was unexpectedly found in the apical third of the canals during treatment, and the tooth was successfully treated by removing the necrotic pulp in the coronal and middle thirds of the canals. Later, Mineral Trioxide Aggregate (MTA) was applied as a filling material.
Case Presentation
A girl aged seven years and ten months was referred to an endodontist at the Lithuania University of Health Sciences, Department of Dental and Oral Pathology by her general dental practitioner. The patient complained of moderate pain in the area of the mandibular right first molar. Her dental history indicated that five months prior, tooth #46 had been treated via the indirect pulp capping method using TheraCal (Bisco Inc.) and Activa BioActive (Pulpdent, Watertown, MA, USA) composite. Two weeks prior, the general dental practitioner had started treatment because of severe pain and intraoral swelling next to #46. The patient was treated with an antibiotic for six days.
Clinical examination found no extraoral or intraoral swelling. Tooth #46 had a temporary restoration in the occlusion surface. The tooth was tender to percussion, and thermal sensibility tests were negative. Periapical radiographs showed that tooth #46 had a periapical radiolucency and immature roots (Figure 1). The tooth was diagnosed with symptomatic apical periodontitis.
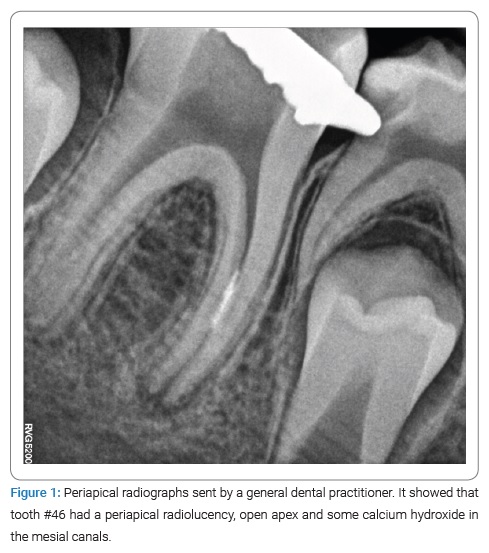
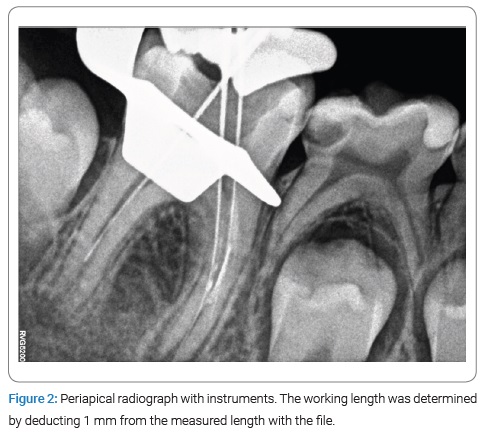
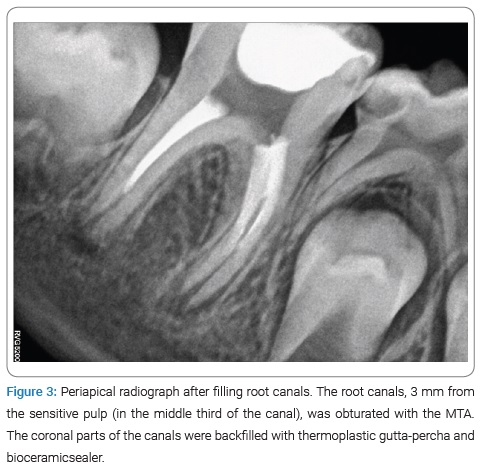
Without administering anesthesia, the tooth was isolated with a rubber dam. The access cavity was prepared with carbide burs under the microscope by removing the temporary filling in the occlusion surface. Necrotic pulp tissues were removed from the coronal part, and irrigation using 0.5% sodium hypochlorite (NaOCl) was performed. A hand #30 K-file was introduced approximately 2/3 of the way into the canal length, as the patient reported discomfort. Intracanal bleeding was observed, the file was withdrawn, and the length of the file was measured. The working length was determined to be 1 mm shorter than the length measured with the file (Figure 2). An apex locator was not used. All three root canals were chemo-mechanically prepared at working lengths (2/3 of the canals)with rotary Protaper Gold (Dentsply, Konstanz, Germany) instruments; the last instrument was F5 (size #50 K-file). NaOCl (0.5%) was used as an intracanal irrigant, and calcium hydroxide (Ultracal; Ultradent Inc., South Jordan, USA) was used for intracanal medication. After two weeks, tooth #46 was reopened and disinfected with 0.5% NaOCl. Final irrigation with 5 mL of 17-% ethylenediaminetetraacetic acid was performed. After drying with paper points, the root canals, 3mm from the sensitive pulp (in the middle third of the canal), was obturated with the MTA (ProRoot MTA, Densply – Sirona, Charlotte, NC, USA). The canal walls were cleaned with wet paper points, the TotalFill BC Sealer (FKG, La Chaux-des-Fonds, Switzerland) was applied, and the coronal parts of the canals were backfilled with Calamus thermoplastic gutta-percha (Densply, Tulsa, OK) (Figure 3). The tooth’s access cavity was etched with 35% phosphoric acid (Scotchbond Etchant, 3M ESPE) for 15s, rinsed with water, and then gently dried with air spray. Two layers of adhesive (3M Bond, USA) were applied with a micro brush, and each layer was dispersed by applying a weak air stream and then cured with a light-emitting diode for 10s. Finally, the restoration of the composite resin (3M ESPE, USA) was applied.
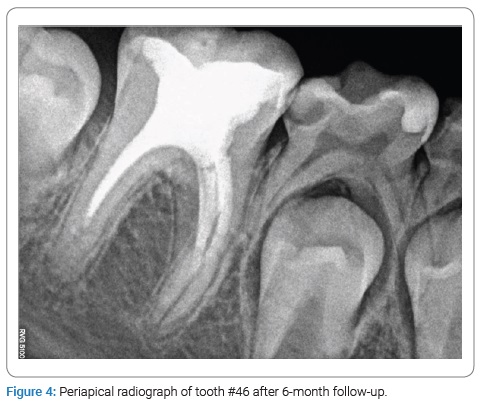
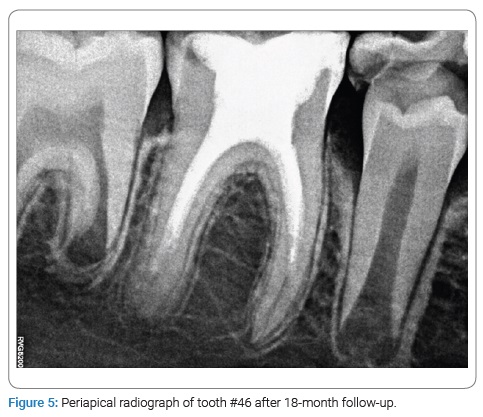
At the 6-month recall, the patient had no symptoms, and no periapical radiolucency was seen. At that time, the roots’ lengths had increased slightly, and pulp canal space was reduced (Figure 4). At the 18-month follow-up, no symptoms of periapical radiolucency appeared. The apical foramen was closed, and the roots’ lengths had increased (Figure 5).
Discussion
This case report was to show a different way of treating immature teeth with partial pulp necrosis. Modified apexogenesis was chosen to save vital pulp tissues and encourage continued root maturation in an immature permanent tooth.
Necrosis and infection in the pulp tissue eventually release proinflammatory mediators, which may diffuse through the vital tissue to cause periapical changes [12]. Although they are microorganisms, their metabolic products, toxins, enzymes, and disintegration products from damaged pulp directly influence periapical tissues [1].
In the present report, the occurrence of acute apical periodontitis symptoms in a tooth with vital pulp tissue may be explained by the large apical foramen, which permits a sufficient blood supply to carry the immune system’s cellular and molecular components to the tissue that is under bacterial insult. Because the pulp tissue located deeper in the canals is inflamed but not infected, cleaning till vital tissues and irrigation with NaOCl removed the infected part and helped the inflamed tissue to return to normal. Histologically, it has been demonstrated that teeth with necrotic pulp in the coronal or in the middle part of canals may contain vital pulp tissue in the apical part of the canal space [13]. Therefore, if the infected part of the pulp is removed and the vital pulp survives, the periapical lesion can heal [4].
In this case, the vital pulp in the middle 1/3 of the canal was diagnosed by mechanical stimulation, which gave a positive sensitivity result and provoked bleeding. This case was treated using apexogenesis principles to save the vitality of a tooth with an open apex. The root can develop to normal length because apexogenesis creates the usual physiological conditions for the root. Root development needs two types of cells: epithelial cells of Hertwig’s root sheath and odontoblasts. The former cells are present at the apical end of the immature root, and they resist destruction, even in the presence of inflammation [5]. The epithelial cells of Hertwig’s root sheath can induce the differentiation of mesenchymal stem cells into odontoblasts that subsequently form the root dentin. These mesenchymal stem cells can predominately come from either the residual pulp tissue of the immature permanent teeth or the apical papilla of the immature permanent teeth (so-called stem cells from the apical papilla [SCAP]) as Sonoyama et al. [14,15] and Huang et al. [16] reported. SCAPs residing in the apical papilla may survive the infection because of their proximity to the periapical tissues. After endodontic disinfection and under the influence of the surviving epithelial cells of Hertwig’s root sheath, SCAPs differentiate into primary odontoblasts to complete the root formation [16].
Of course, 18-month follow-up is not enough, and continuous check-ups are needed because of the possibility of failure or complication.
In conclusion, immature teeth should be treated conservatively to enable Hertwig’s sheath to complete physiological root development.
Conclusion
In the presented case, endodontic treatment was based on saving vital pulp tissues and encouraging continued root maturation in an immature permanent tooth. This case report showed a different way of treating immature teeth by applying apexogenesis principles.
Acknowledgments
Published with the consent of the patient.
Author Contribution: Mickeviciene L: performed the treatment and performed the follow-up; Mickeviciene L and Venskutonis T: led the writing.
Ethical Approval: Written informed consent on ethics has been obtained.
Conflict of Interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Informed consent was obtained for this publication.
References
- Lin L, Langeland K. Light and electron microscopic study of teeth with carious pulp exposures. Oral Surg. 1981;51:292–316.
- Holland R, Valle GF, Taintor JF, Ingle JI. Influence of bony resorption on endodontic treatment. Oral Surg Oral Med Oral Pathol. 1983;55:191–203.
- Russo MC, Holland R, de Souza V. Radiographic and histological evaluation of the treatment of inflamed dental pulps. Int Endod J. 1982;15(3):137–142.
- Huang GTJ, Sonoyama W, Liu Y, Liu H, Wang S, Shi S. The hidden treasure in apical papilla: the potential role in pulp/dentin regeneration and bioroot engineering. J Endod. 2008;34(6):645–651.
- Lin L, Shovlin F, Skribner J, Langeland K. Pulp biopsies from the teeth associated with periapical radiolucency. J Endod. 1984;10(9):436–448.
- Iwaya SI, Ikawa M, Kubota M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol. 2001;17(4):185–187.
- Langeland K, Langeland LK, Anderson DM. Corticosteroids in dentistry. Int Dent J. 1977;27(3):217–251.
- Murray PE. Review of guidance for the selection of regenerative endodontics, apexogenesis, apexification, pulpotomy, and other endodontic treatments for immature permanent teeth. Int Endod J. 2022;00;1–12.
- Mousivand S, Sheikhnezami M, Moradi S, Koohestanian N, Jafarzadeh H. Evaluation of the outcome of apexogenesis in traumatised anterior and carious posterior teeth using mineral trioxide aggregate: a 5-year retrospective study. Aust Endod J. 2022;48(3):1–7.
- Ricucci D, Langeland K. Apical limit of root canal instrumentation and obturation, part 2. A histological study. Int Endod J. 1998;31(6):394–409.
- Goldstein S, Sedaghat-Zandi A, Greenberg M, Friedman S. Apexification & apexogenesis. N Y State Dent J. 1999;65(5):23–25.
- Ricucci D, Siqueira JF Jr. Endodontology: an integrated biological and clinical view. London: Quintessence Publishing; 2013.
- Saad AY. Calcium hydroxide and apexogenesis. Oral Surg Oral Med Oral Pathol. 1988;66(4):499–501.
- Chueh LH, Huang GT-J. Immature teeth with periradicular periodontitis or abscess undergoing apexogenesis: a paradigm shift. J Endod. 2006;32(12):1205–1213.
- Sonoyama W, Liu Y, Yamaza T, Tuan RS, Wang S, Shi S, et al. Characterization of the apical papilla and its residing stem cells from human immature permanent teeth: a pilot study. J Endod. 2008;34(2):166–171.
- Tsukiboshi M, Ricucci D, Jr Siqueira JF. Mandibular premolars with immature roots and apical periodontitis lesions treated with Pulpotomy: report of 3 cases. J Endod. 2017;43(9S):S65–S74.
Keywords
Pulpotomy; Immature teeth; MTA plug; Pulp regeneration; Apexogenesis; Permanent teeth
Cite this article
Mickeviciene L, Venskutonis T. Treatment of a partially necrotic molar tooth by applying apexogenesis principles. Clin Case Rep J. 2023;4(2):1–4.
Copyright
© 2023 Mickeviciene L. This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY-4.0).





