Abstract
Introduction: Guillain-Barré Syndrome (GBS) is an acute, immune-mediated peripheral neuropathy typically triggered by antecedent infections. While several pathogens have been implicated, Methicillin-Resistant Staphylococcus aureus (MRSA) is a rarely reported cause.
Case Presentation: We describe a 51-year-old man who developed progressive bilateral lower extremity weakness and sensory changes two months after treatment for MRSA cellulitis. Diagnosis was confirmed by cerebrospinal fluid analysis demonstrating albuminocytologic dissociation and electromyography showing demyelinating polyneuropathy. The patient was treated with intravenous immunoglobulin and plasma exchange with limited early improvement.
Conclusion: This case highlights MRSA as a potential, though rare, trigger for GBS and emphasizes the importance of considering GBS in patients presenting with neurological symptoms following MRSA infections.
Introduction
Guillain-Barré Syndrome (GBS) is an acute, immune-mediated polyradiculoneuropathy that often follows a preceding infection. While Campylobacter jejuni is the most frequently implicated pathogen, numerous other infectious and non-infectious triggers have been identified [2–4]. Viral antecedents include Cytomegalovirus (CMV), Epstein–Barr virus (EBV), Zika virus, Varicella-Zoster Virus (VZV), Hepatitis E Virus (HEV), dengue virus, and SARS-CoV-2. Bacterial causes such as Mycoplasma pneumoniae, Bartonella henselae (cat scratch disease), and Brucella spp. (neurobrucellosis) have also been reported. In rare cases, GBS has followed non-infectious events such as vaccinations or even envenomation, including snake bites [11]. These diverse triggers suggest a shared pathophysiologic mechanism likely involving molecular mimicry, leading to immune-mediated damage to peripheral nerves [1–3].
Case Presentation
A 51-year-old Caucasian man with diabetes mellitus, pulmonary hypertension, and obstructive sleep apnea presented with five days of progressive bilateral lower extremity weakness, paresthesias, and difficulty ambulating. Symptoms steadily worsened during this time. There were no signs of cranial nerve involvement, respiratory compromise, or autonomic instability.
Two months before presentation, the patient completed a six-week course of intravenous antibiotics and underwent skin grafting for MRSA cellulitis of the posterior neck [5]. Initial rehabilitation was uneventful. However, soon after completing rehabilitation, he developed neurological symptoms, which progressed over a period of five days prior to admission.
On physical examination, he was afebrile and hemodynamically stable. Neurological examination revealed 5/5 strength in the upper extremities and 3/5 in the lower extremities, with diminished deep tendon reflexes bilaterally. Sensory examination demonstrated decreased light touch and pinprick sensation in the lower limbs. Cranial nerves were intact, and no respiratory compromise was noted.
Investigations included:
Cerebrospinal fluid (CSF) analysis: Elevated protein (117 mg/dL) with normal white blood cell count (2 cells/mm³), consistent with albuminocytologic dissociation [1].
MRI Brain: In addition to the CSF findings, imaging revealed non-specific T2 hyperintensities in the left parietal-occipital region without evidence of acute ischemia.
Electromyography/Nerve Conduction Studies (EMG/NCS): Furthermore, these studies showed findings consistent with Acute Inflammatory Demyelinating Polyneuropathy (AIDP) [6].
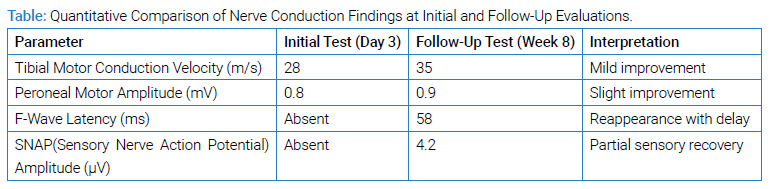
Electrodiagnostic Timing and Correlation: Initial EMG/NCS was performed on hospital day 3, showing reduced motor amplitudes, slowed conduction velocities, and absent F-waves — consistent with demyelinating polyneuropathy (AIDP subtype of GBS). A follow-up study was conducted at week 8, showing partial recovery of motor amplitudes, reappearance of F-waves, and improved conduction velocities, corresponding clinically with improved lower limb strength and gait stability [7]. Sensory responses remained diminished, consistent with residual symptoms. To further evaluate the underlying etiology, extensive infectious testing was pursued.
Extensive infectious testing, including enterovirus, Lyme disease, herpes simplex virus, cytomegalovirus, West Nile virus, syphilis, and varicella-zoster virus PCRs, returned negative. The patient was treated with Intravenous Immunoglobulin (IVIG) at 400 mg/kg/day for five days, initiated on hospital day 4, followed by five sessions of plasma exchange over the subsequent two weeks [8,9]. At hospital discharge (week 3), improvement was limited, necessitating inpatient rehabilitation. Follow-up studies at week 8 confirmed AIDP, and the patient demonstrated gradual recovery over subsequent months (Table).
Discussion
GBS is an acute inflammatory disorder defined by progressive, symmetrical weakness and areflexia. While classically associated with Campylobacter jejuni and various viral infections, it increasingly appears that a broad spectrum of antecedent pathogens can be implicated [2–4]. MRSA frequently causes cutaneous and soft tissue infections, yet reports of its association with GBS remain rare [5].
Extending from the clinical picture already described, the proposed mechanism involves molecular mimicry. In this process, immune responses targeting bacterial antigens cross-react with gangliosides in peripheral nerves [6]. Additionally, the temporal association between MRSA cellulitis treatment and neurological symptom onset suggests a causal relationship. Although similar cases have been reported previously, the association remains rare and underrecognized [5].
Given these considerations, prompt GBS diagnosis is essential. Early IVIG or plasma exchange can improve outcomes. In our patient, standard therapy was initiated, but the initial response was limited, highlighting variability in GBS recovery [10]. This case expands the range of recognized triggers for GBS. Building on the prior case details, it also highlights the importance of considering MRSA as a potential antecedent infection in patients presenting with post-infectious neuropathy.
Conclusion
This case demonstrates a rare instance of GBS following MRSA cellulitis and reinforces the need to maintain a broad differential diagnosis in patients with progressive weakness after infection. Early recognition and intervention remain key to optimizing outcomes.
Acknowledgments
Author Contributions: AA drafted the initial manuscript. RB and JM contributed to clinical data collection and manuscript revision. PJ provided senior supervision and final approval. All authors read and approved the final manuscript.
Consent: Written informed consent was obtained from the patient for publication of this case report.
Data Availability Statement: Data sharing is not applicable to this article as no datasets were generated or analyzed during the current study.
Conflict of Interest
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Informed consent was obtained for this publication.
Keywords
Guillain-Barré syndrome; Methicillin-resistant Staphylococcus aureus; MRSA; Neuropathy; Intravenous immunoglobulin; Plasma exchange; Casereport
Cite this article
Akinwumiju A, Balsiger RW, Mahajan JP, Janda P. Guillain-Barré Syndrome Following Methicillin-Resistant Staphylococcus aureus Cellulitis: A Case Report. Clin Case Rep J. 2026;7(1):1–3.
Copyright
© 2026 Akinyemi Akinwumiju. This is an open access article distributed under the terms of the Creative Commons Attribution 4.0 International License (CC BY-4.0).

